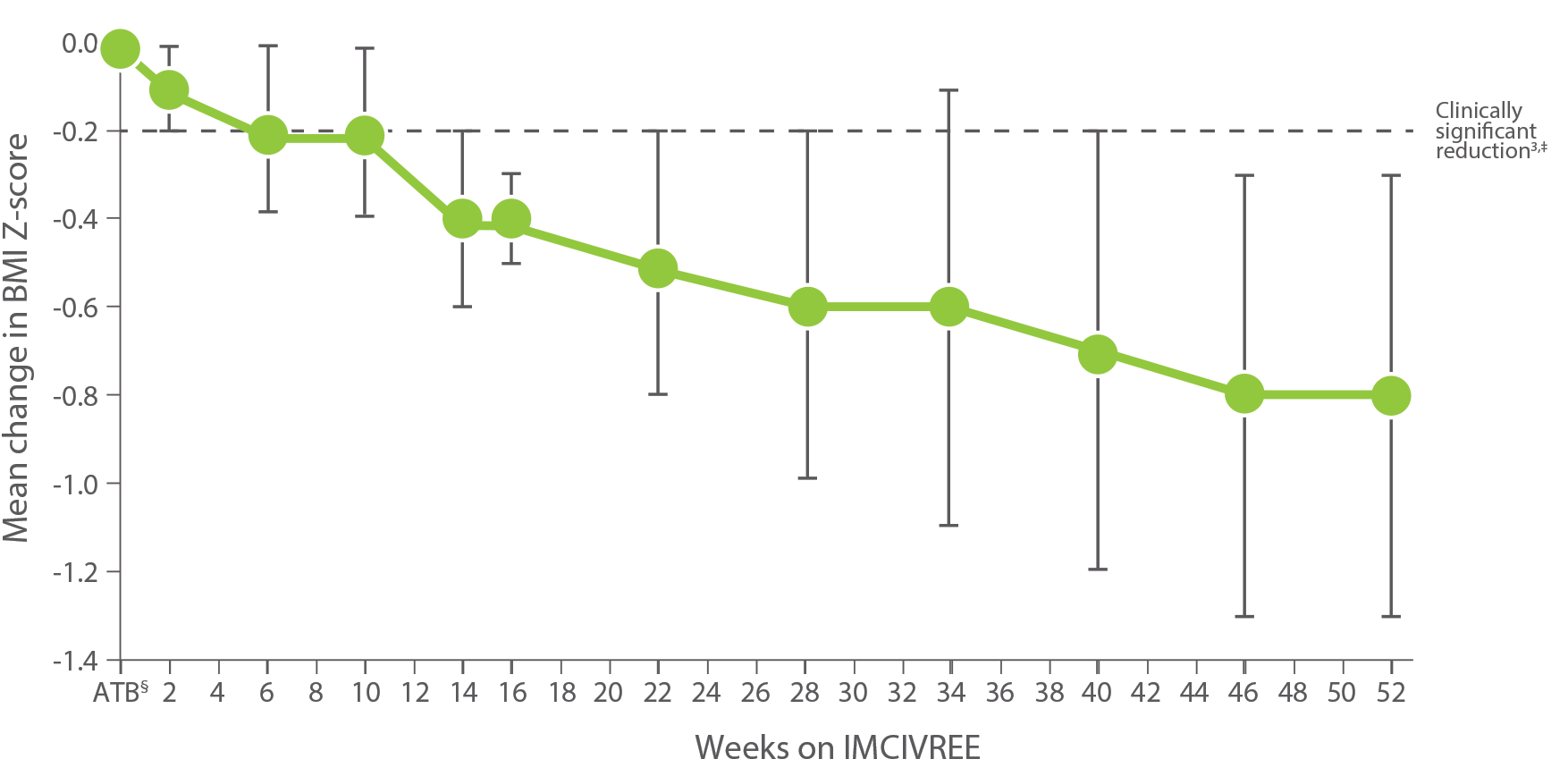
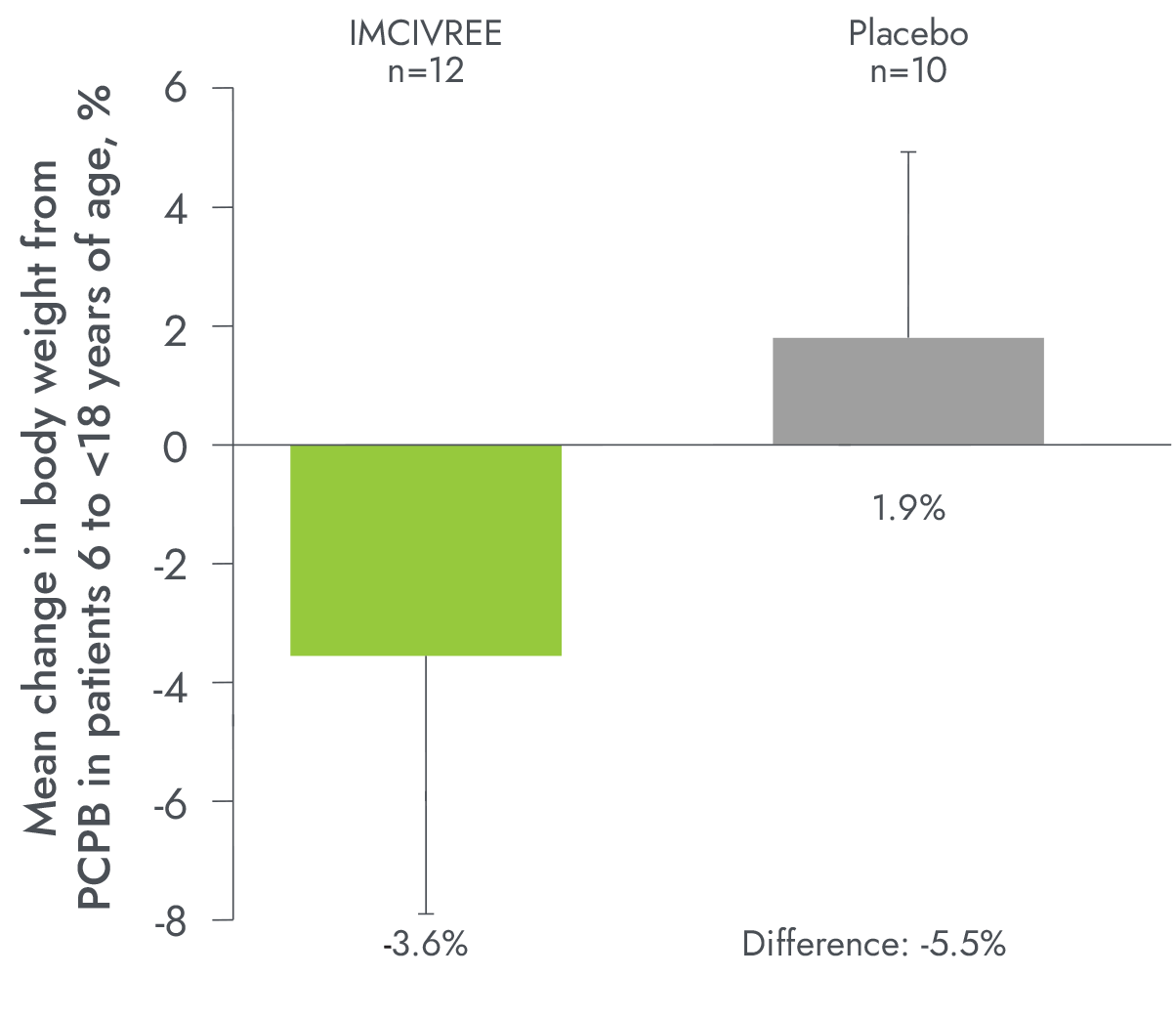
IMCIVREE delivered early, significant, and sustained weight reduction1-3
Change in BMI Z-score from baseline in patients 6 to <18 years of age after 52 weeks (n=14)1
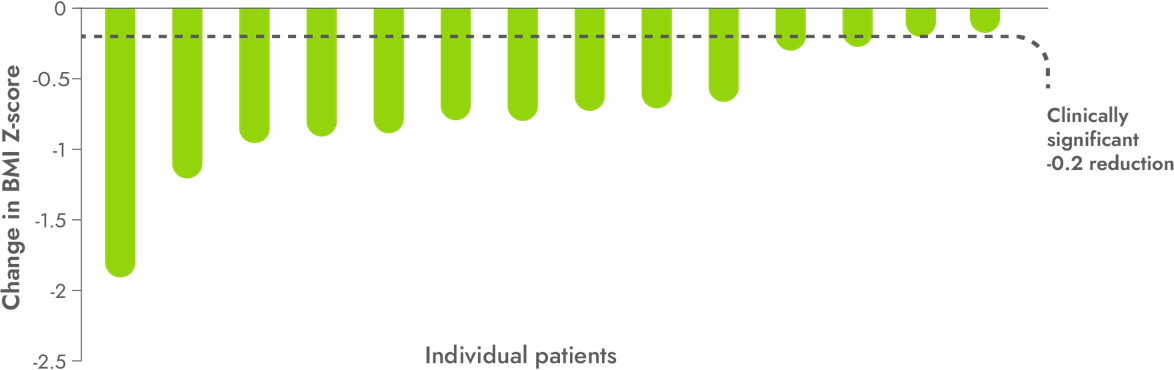
86%
of patients achieved a clinically significant ≥0.2 reduction* in BMI Z-score1,3
- 100% of patients <12 years of age achieved a clinically significant ≥0.2 reduction in BMI Z-score (n=3)1,3
*BMI Z-score was an exploratory endpoint used to measure the reduction in BMI in children. A clinically significant reduction is generally considered a ≥0.2 reduction in BMI Z-score. A 0.2 reduction is comparable to weight loss of approximately 5%.3
Patients were not required to change their diet or exercise routine1
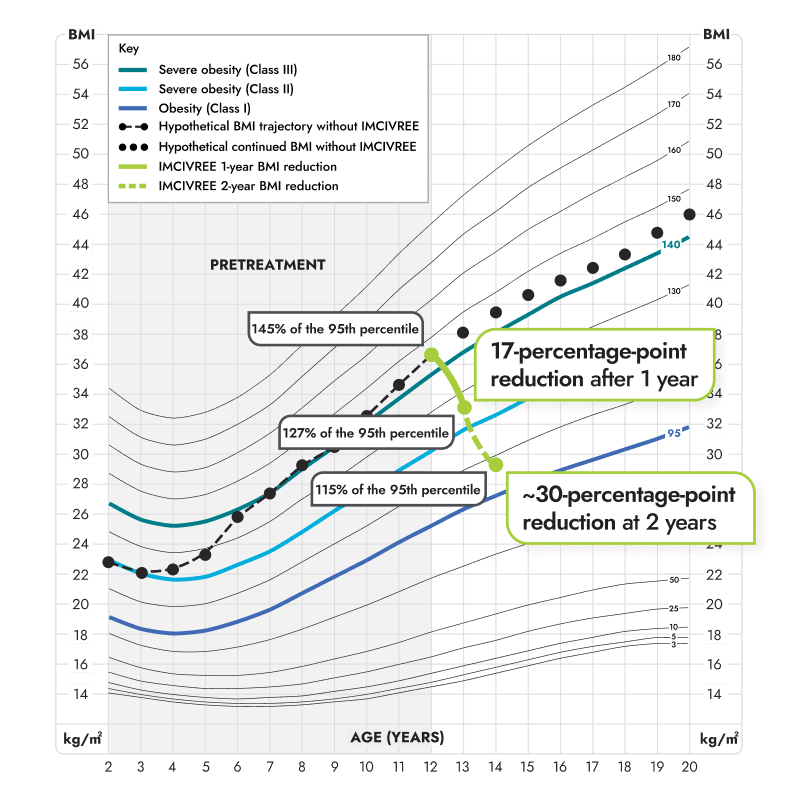
Figure modeled after Gulati AK, Kaplan DW, Daniels SR. Clinical tracking of severely obese children: a new growth chart. Pediatrics. 2012;130(6):1136-1140.
*Growth chart is based on females 2 to 20 years of age and is for illustrative purposes only.
References: 1. Data on file. Rhythm Pharmaceuticals, Inc. Boston, MA. 2. Haqq AM et al. Lancet Diabetes Endocrinol. 2022;10(12):859-868. doi:10.1016/S2213-8587(22)00277-7. Supplemental appendix available at: https://www.thelancet.com/journals/landia/article/PIIS2213-8587(22)00277-7/fulltext. 3. Grossman DC et al; US Preventive Services Task Force. JAMA. 2017;317(23):2417-2426. 4. IMCIVREE [prescribing information]. Boston, MA. Rhythm Pharmaceuticals, Inc. 5. Argente J et al. Endocrine Society Annual Meeting. Poster ODP606. June 11-14, 2022. 6. Vanderwall C et al. BMC Pediatrics. 2018;18(1):187. 7. Defining child BMI categories. Centers for Disease Control and Prevention. Accessed August 11, 2023. https://www.cdc.gov/obesity/basics/childhood-defining.html. 8. Centers for Disease Control and Prevention. 2000 CDC Growth Charts for the United States: Methods and Development. Accessed August 11, 2023. https://www.cdc.gov/nchs/data/series/sr_11/sr11_246.pdf. 9. Kelly AS et al. Circulation. 2013;128(15):1689-1712. 10. Gulati AK et al. Pediatrics. 2012;130(6):1136-1140. 11. Racette SB et al. BMC Pediatr. 2017;17(1):130. 12. Argente J et al. The Pediatric Endocrine Society Annual Meeting. Poster 155. April 28-May 1, 2022.