Indication
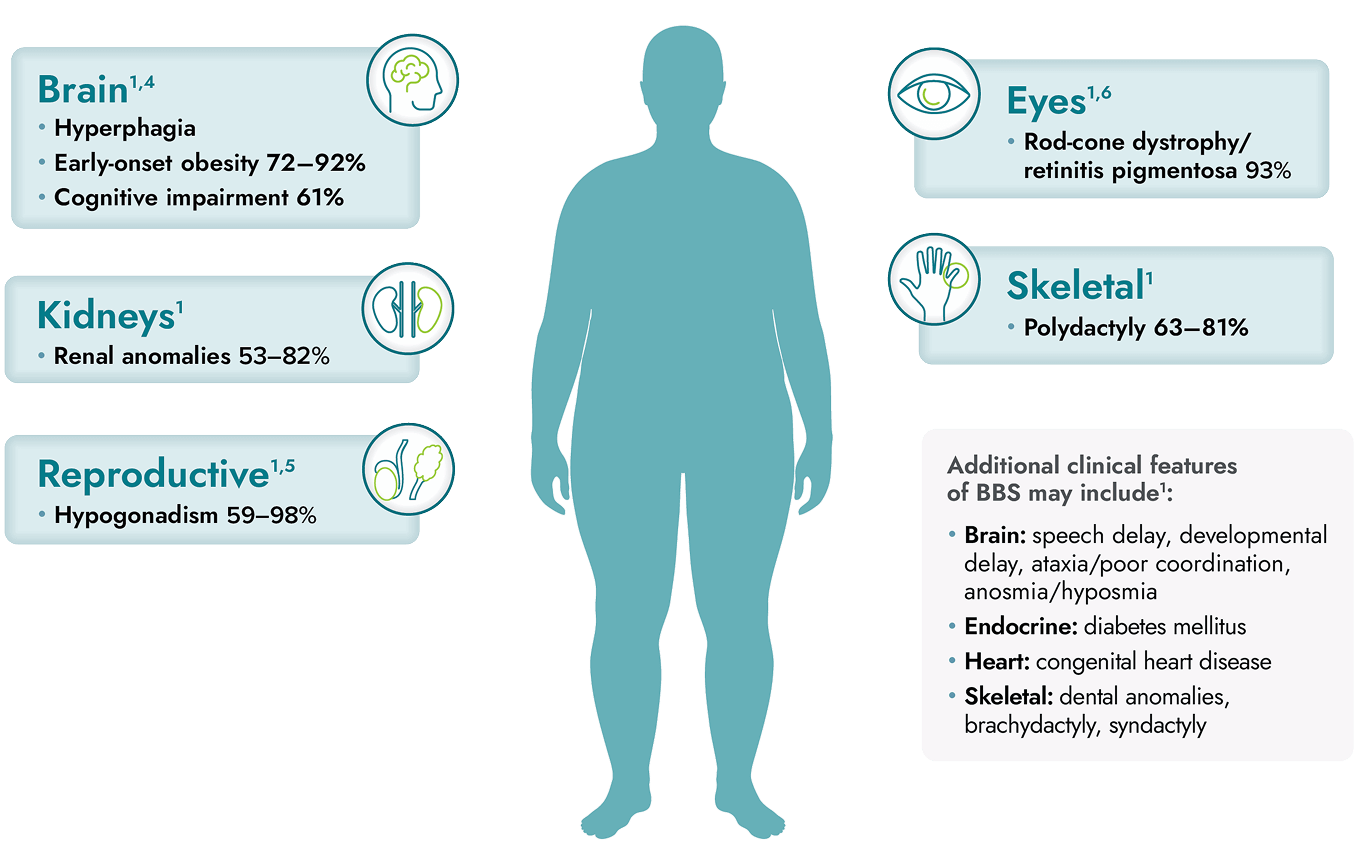
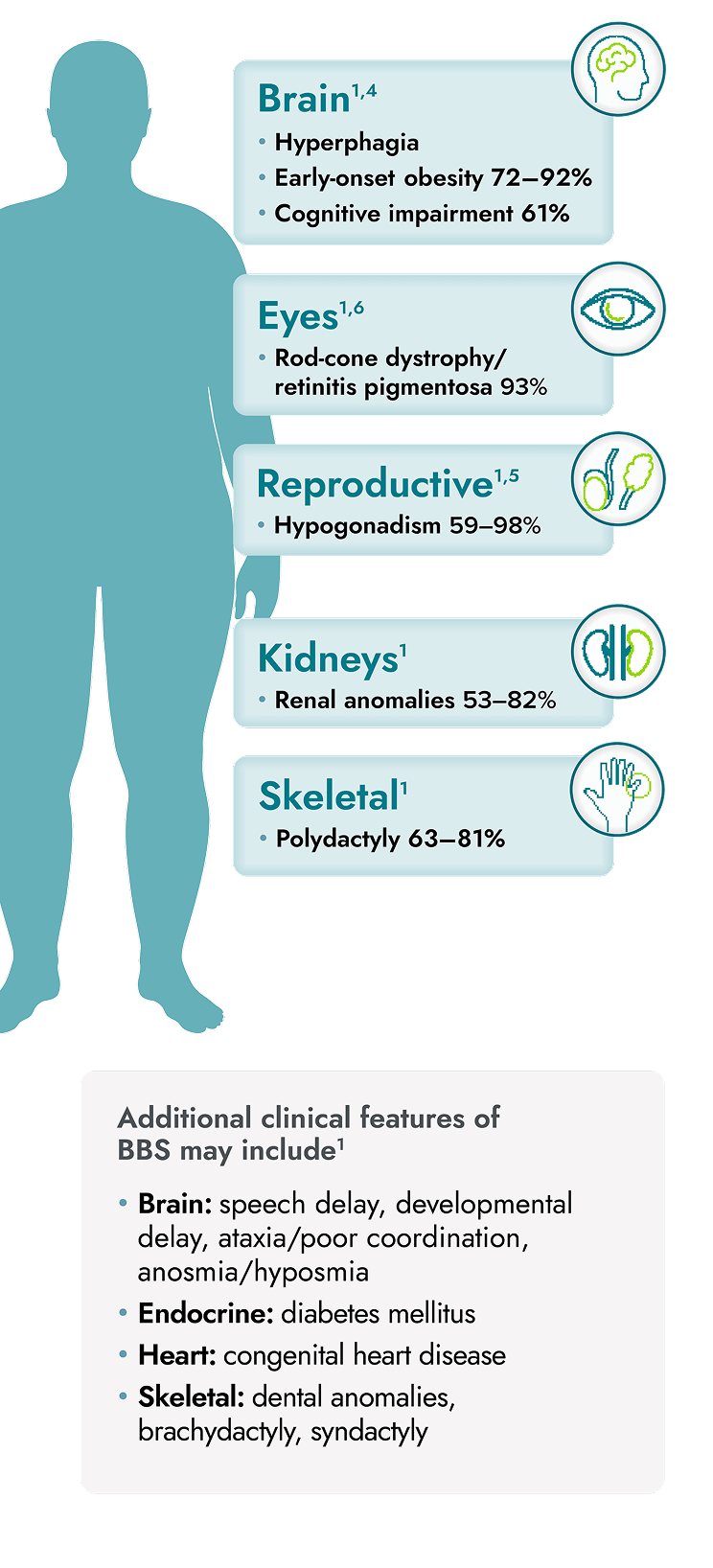
IMCIVREE is indicated to reduce excess body weight and maintain weight reduction long term in adults and pediatric patients aged 2 years and older with syndromic or monogenic obesity due to Bardet-Biedl syndrome (BBS).
Limitations of Use
IMCIVREE is not indicated for the treatment of patients with the following conditions as IMCIVREE would not be expected to be effective:
- Other types of obesity not related to BBS or other FDA-approved indications for IMCIVREE, including obesity associated with other genetic syndromes and general (polygenic) obesity
Important Safety Information
CONTRAINDICATIONS
Prior serious hypersensitivity to setmelanotide or any of the excipients in IMCIVREE. Serious hypersensitivity reactions (e.g., anaphylaxis) have been reported.
WARNINGS AND PRECAUTIONS
Disturbance in Sexual Arousal: Spontaneous penile erections in males and sexual adverse reactions in females have occurred. Inform patients that these events may occur and instruct patients who have an erection lasting longer than 4 hours to seek emergency medical attention.
Depression and Suicidal Ideation: Depression, suicidal ideation, and depressed mood have occurred. Monitor patients for new onset or worsening depression or suicidal thoughts or behaviors. Consider discontinuing IMCIVREE if patients experience suicidal thoughts or behaviors, or clinically significant or persistent depression symptoms occur.
Hypersensitivity Reactions: Serious hypersensitivity reactions (e.g., anaphylaxis) have been reported. If suspected, advise patients to promptly seek medical attention and discontinue IMCIVREE.
Skin Hyperpigmentation, Darkening of Pre-existing Nevi, and Development of New Melanocytic Nevi: Generalized or focal increases in skin pigmentation, darkening of pre-existing nevi, development of new melanocytic nevi and increase in size of existing melanocytic nevi have occurred. Perform a full body skin examination prior to initiation and periodically during treatment to monitor pre-existing and new pigmentary lesions.
Risk of Serious Adverse Reactions Due to Benzyl Alcohol Preservative in Neonates and Low Birth Weight Infants: IMCIVREE is not approved for use in neonates or infants. Serious and fatal adverse reactions including “gasping syndrome” can occur in neonates and low birth weight infants treated with benzyl alcohol-preserved drugs.
ADVERSE REACTIONS
- Most common adverse reactions (incidence ≥20%) included skin hyperpigmentation, injection site reactions, nausea, headache, diarrhea, abdominal pain, vomiting, depression, and spontaneous penile erection
USE IN SPECIFIC POPULATIONS
Treatment with IMCIVREE is not recommended when breastfeeding. Discontinue IMCIVREE when pregnancy is recognized unless the benefits of therapy outweigh the potential risks to the fetus.
To report SUSPECTED ADVERSE REACTIONS, contact Rhythm Pharmaceuticals at 833-789-6337 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Please see the full Prescribing Information for additional Important Safety Information.