How did IMCIVREE help reduce measures of weight and hunger in the clinical trials?
In clinical studies, IMCIVREE reduced weight and hunger for people with obesity due to LEPR deficiency
WEIGHT
STUDY 2
Children & adults 6 years and older with obesity due to LEPR deficiency
5 out of 11 people lost at least 10% of their body weight at 1 year
*Participants who experienced weight loss of 5 kg/11 lb (or 5% if their starting weight was less than 100 kg/220 lb) during the first 10 weeks.
HUNGER
STUDY 2
Children & adults 6 years and older with obesity due to LEPR deficiency
After 1 year, participants (n=8) experienced a decrease from 7.0 to 4.4 in the median† daily hunger score.
Changes in hunger were evaluated using a questionnaire that was completed each day for 1 year, by participants who were 12 years of age or older. The questionnaire measured hunger using a score ranging from 0 ("not hungry at all") to 10 ("hungriest possible").
†A median is the middle value of a set of data that have been put into numerical order. The median is the value that divides the data into 2 halves.
Impact of stopping treatment
Treatment was stopped for a period of time to see how it impacted weight and hunger.
Over this withdrawal period:
- Weight increased; when the withdrawal period ended and treatment was restarted, weight loss continued
- Hunger scores generally worsened; hunger scores improved once treatment was restarted
In a clinical study, IMCIVREE reduced weight in young children with obesity due to POMC or LEPR deficiency.
WEIGHT
STUDY 3
Young children ages 2 to less than 6 years old with obesity due to POMC or LEPR deficiency*
After 1 year of treatment with IMCIVREE:
Patients with POMC deficiency had a mean reduction in BMI of
~34%
Patients with LEPR deficiency had a mean reduction in BMI of
~13%
100%
young children (3 out of 3) with POMC deficiency
75%
young children (3 out of 4) with LEPR deficiency
had a meaningful reduction in BMI Z-score over 1 year
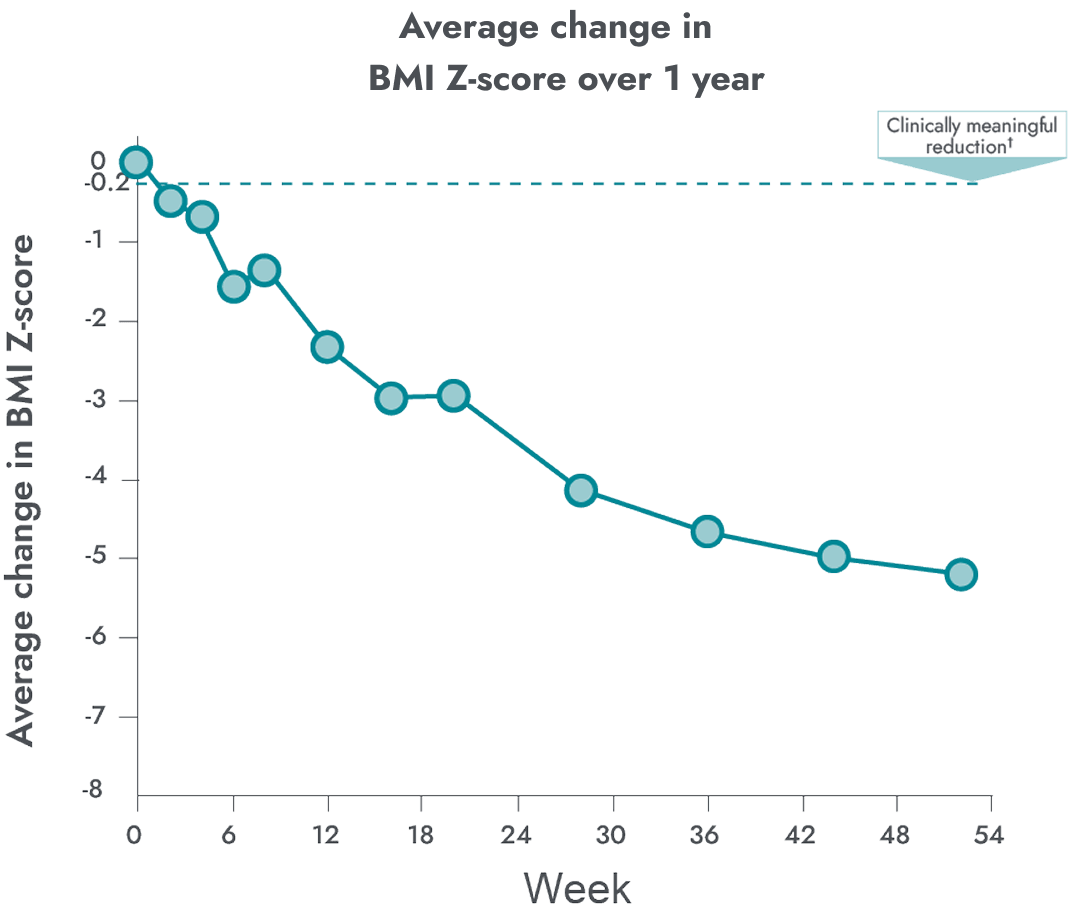
A clinically meaningful 5.19 mean reduction in BMI Z-score at week 52
*Patients with PCSK1 deficiency were eligible but none enrolled in the trial.
†A clinically meaningful reduction is generally considered a ≥0.2 reduction in BMI Z-score. A 0.2 reduction is comparable to weight loss of approximately 5%.
A lot has changed, and when I look at the hunger today, I’m assuming it’s on the same level as other people.
— Person who is living with POMC deficiency
I’m very satisfied I’ve lost a lot of weight, and I have a lot more energy. It’s nice to see that someone is paying attention to this, and there’s a medication for this, that they’re deploying people to deal with this problem, when people used to have to go without care.
— Person who is living with POMC deficiency
Now, I really feel it. My stomach growls and I notice that I am hungry. Experiencing both fullness and hunger is something I have never felt before. It's funny.
— Person who is living with LEPR deficiency